UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
PURSUANT TO SECTION 13 OR 15(d)
OF THE SECURITIES EXCHANGE ACT OF 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in charter)
| (State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(I.R.S. Employer Identification No.) |
(Address of Principal Executive Offices, including Zip Code)
(Registrant’s telephone number, including area code)
(Former name or former address, if changed since last report.)
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading |
Name of each exchange on which registered | ||
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 1.01. | Entry into a Material Definitive Agreement. |
On September 19, 2025, Cyclerion Therapeutics, Inc. (the “Company”) and the Massachusetts Institute of Technology (“MIT”) entered into a Patent License Agreement (the “License Agreement”) pursuant to which MIT granted to the Company an exclusive worldwide license to develop and commercialize products using certain technology for the treatment of neuropsychiatric disorders, such as depression, in humans.
Under the terms of the License Agreement, the Company will pay to MIT a nominal upfront payment, and MIT will be eligible to receive up to $4.4 million upon the achievement of certain development, regulatory and sales milestone payments. MIT will also receive tiered royalties in a range of percentages in the low single digits based on future net sales of licensed products as set forth in the License Agreement. Further, the Company is required to pay MIT varying percentages of income received as consideration for any sublicenses granted pursuant to the License Agreement depending on the circumstances of the sublicense and the development milestones of sublicensed products.
The Company will be responsible for developing and seeking regulatory approval of the licensed products. Following regulatory approval, the Company will be responsible for commercializing the licensed products in the jurisdiction in which regulatory approval is obtained. MIT will be responsible for filing, prosecuting and maintaining all of the licensed patent rights in cooperation with the Company.
The term of the License Agreement will expire in its entirety upon the expiration of certain patent rights for the licensed patents, unless earlier terminated by the parties in accordance with the terms of the License Agreement.
The foregoing description of the terms of the License Agreement is not complete and is qualified in its entirety by reference to the text of the License Agreement, a copy of which the Company intends to file as an exhibit to its Quarterly Report on Form 10-Q for the period ended September 30, 2025.
| Item 7.01 | Regulation FD Disclosure |
On September 23, 2025, the Company issued a press release announcing its entry into the License Agreement and relaunch as a neuropsychiatric company.
In addition, the Company has prepared an investor presentation with respect to the License Agreement and relaunch as a neuropsychiatric company.
A copy of the press release and investor presentation are attached to this Current Report on Form 8-K as Exhibit 99.1 and Exhibit 99.2, respectively, and incorporated herein by reference. The exhibits furnished under Item 7.01 of this Current Report on Form 8-K shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Exchange Act or the Securities Act, regardless of any general incorporation language in such filing.
| Item 9.01. | Financial Statements and Exhibits. |
(d) Exhibits
| Exhibit No. |
Description | |
| 99.1 | Press release, dated September 23, 2025 | |
| 99.2 | Investor Presentation, dated September 24, 2025. | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL). | |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| CYCLERION THERAPEUTICS, INC. | ||||||
| Date: September 23, 2025 | By: | /s/ Regina Graul, Ph.D. | ||||
| Name: | Regina Graul, Ph.D. | |||||
| Title: | President and Chief Executive Officer | |||||
Exhibit 99.1

Cyclerion Announces Transformational Relaunch as a Neuropsychiatric Company
– Cyclerion Relaunches, Advancing a Phase 2–Ready Program for Millions Living with Treatment-Resistant Depression Through MIT License Agreement –
– Webcast to be held September 24, 2025 at 10:30 a.m. ET –
CAMBRIDGE, Mass., September 23, 2025 (GLOBE NEWSWIRE) – Cyclerion Therapeutics, Inc. today announced that it has entered into a licensing agreement with the Massachusetts Institute of Technology (“MIT”), securing the intellectual property that will serve as the cornerstone of its strategic relaunch. This agreement marks a pivotal milestone in Cyclerion’s transformation into a new, innovation-driven company focused on delivering potential novel, improved or first-in-class therapies for neuropsychiatric conditions with large unmet needs, beginning with treatment-resistant depression (“TRD”).
“This relaunch represents a pivotal moment for Cyclerion Therapeutics as we refocus our efforts on advancing breakthrough neuropsychiatric treatments for patients who have limited therapeutic options today,” said Dr. Errol DeSouza, a leader in the advancement of treatments for Central Nervous Disorders and Chairperson of Cyclerion’s Board of Directors. “Our board remains committed to partnering with management to support the strategic vision and ensure we have the resources necessary to develop innovative therapies that can meaningfully improve outcomes for individuals suffering from serious neuropsychiatric conditions. We believe this new chapter positions the company to make a significant impact in this underserved therapeutic area while creating sustainable value for all stakeholders.”
Cyclerion’s lead program leverages common anesthetic agents and a proprietary, tech-driven system to resynchronize communication between key brain regions and restore functional connectivity in patients with TRD. For the 3 million Americans living with TRD, this approach could deliver a long-awaited alternative to current limited treatment options.
“Cyclerion’s approach to drug development is grounded in compelling science and clinical precedent,” said Steven Hyman, M.D., Director of the Program in Brain Health at the Broad Institute and member of Cyclerion’s Board of Directors. “By leveraging known anesthetic mechanisms and pairing them with intelligent delivery systems, Cyclerion is pioneering a new class of neuropsychiatric therapies that could offer meaningful relief to patients with treatment-resistant depression.”
Cyclerion’s relaunch is supported by a world-class team of experts in neuropsychiatry, anesthesiology, machine learning and therapeutic delivery. The Company’s operating model is designed to maximize value creation and advance programs through de-risked inflection points. A Phase 2 proof-of-concept trial for its lead program is expected to initiate in 2026, with an initial data set anticipated in 2027.
In addition to its lead program, Cyclerion is actively exploring expansion into other neuropsychiatric diseases. The company’s strategic vision includes building a pipeline of novel, improved or first-in-class therapies that combine clinical efficacy with commercial differentiation.
“This agreement marks the launch of a new era at Cyclerion,” said Regina Graul, Ph.D., Chief Executive Officer of Cyclerion. “We are building a company that combines the rigor of a leading biopharma with the agility of a startup. For patients who have exhausted traditional options and for those underserved by current treatments, our foundational therapeutic candidate could represent the future of care in treatment-resistant depression. We believe Cyclerion will lead the way in advancing precision neuropsychiatric solutions beyond TRD—potentially bringing forward additional therapies that are safe, effective and accessible.”
Webcast Information
Cyclerion will make a recorded webcast presentation available on its website tomorrow, September 24, 2025 at 10:30am Eastern Time to discuss this milestone and the Company’s future plans. To access the webcast, please visit the “Events” page of the Cyclerion website at www.cyclerion.com. The replay will be available after posting and will remain archived for one year.
For more information about Cyclerion, please visit https://www.cyclerion.com/ and follow us on X (@Cyclerion) and LinkedIn (www.linkedin.com/company/cyclerion).
About Cyclerion Therapeutics
Cyclerion Therapeutics, Inc. (“Cyclerion” or the “Company”) is a clinical-stage biopharmaceutical company focused on building a new pipeline of therapeutics for neuropsychiatric diseases, with an individualized therapy for treatment-resistant depression (“TRD”) as its foundational product candidate. Given the significant unmet medical need in TRD, the program’s stage of clinical development, and the strong commercial opportunity, the Company believes this program is well positioned to serve as the cornerstone of its future growth.
Cyclerion became an independent, publicly traded company on April 1, 2019, following a tax-free spin-off from Ironwood Pharmaceuticals, Inc. The Company was originally established to develop novel soluble guanylate cyclase (sGC) stimulators for serious diseases in both the central nervous system (CNS) and the periphery. As part of its strategic shift toward neuropsychiatry, Cyclerion sold zagociguat and CY3018 to Tisento in 2023, out-licensed praliciguat in 2021, and entered into a non-binding license option agreement for olinciguat in 2024. The Company continues to leverage these legacy sGC assets with the goal of generating revenues to help fund its strategic pipeline in neuropsychiatry.
Forward Looking Statement
Certain matters discussed in this press release are “forward-looking statements”. We may, in some cases, use terms such as “believes,” “potential,” “anticipates,” “expects,” “plans,” “could,” “will,” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. These statements involve risks, uncertainties and other factors that may cause actual results, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. We caution you that these statements are based on a combination of facts and factors currently known by us and our projections of the future, about which we cannot be certain. Forward-looking statements in this press release include, but are not limited to, statements about our ability to develop product candidates and the timing of related milestones. We cannot assure you that the forward-looking statements in this press release will prove to be accurate. Furthermore, if the forward-looking statements prove to be inaccurate, the inaccuracy may be material. Some of the key factors that could cause actual performance and results to differ materially from those projected or suggested in the forward- looking statements include, but are not limited to, the substantial doubt regarding the our ability to continue as a going concern, our ability to raise additional funding, our ability to enroll patients in future clinical studies, our ability to obtain regulatory approval for our product candidates, unanticipated changes to our nonclinical or clinical study protocols due to regulatory reasons or unanticipated events, which could lead to increased costs to us and could delay our development timeline, our reliance on third parties to conduct clinical studies and to manufacture drug supplies for our product candidates, our ability to adequately protect our intellectual property, and the other important risk factors discussed under the heading “Risk Factors” in our Annual Report on Form 10-K filed with the SEC on March 5, 2024 as well as other risks and uncertainties which may be described in any subsequent quarterly report on Form 10-Q filed by the Company and the other reports the Company files with the SEC. In light of the significant uncertainties in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified time frame, or at all. The forward-looking statements are made only as of the date of this press release, and the Company undertakes no obligation to publicly update such forward-looking statements to reflect subsequent events or circumstances.
Contacts
Investor & Media Relations Email:
IR@cyclerion.com


Exhibit 99.2 Cyclerion: Pioneering a New Era in Neuropsychiatric Therapies Investor Call th September 24 , 2025, 10:30am EST

2 Legal Disclaimer This presentation has been prepared by Cyclerion Therapeutics, Inc. and its consolidated subsidiaries (“the Company,” “Cyclerion,” “we,” “us,” and “our”). Certain matters discussed in this presentation are “forward-looking statements.” We may, in some cases, use terms such as “potential,” “may,” “expects,” “plans,” “could,” “opportunity,” or other words that convey uncertainty of future events or outcomes to identify these forward-looking statements. These statements involve risks, uncertainties and other factors that may cause actual results, performance or achievements to be materially different from the information expressed or implied by these forward-looking statements. We caution you that these statements are based on a combination of facts and factors currently known by us and our projections of the future, about which we cannot be certain. Forward-looking statements in this presentation include, but are not limited to, statements about the timing of development and commercialization of our product candidates, our ability to develop product candidates and the timing of related milestones, creation of shareholder value, adoption of our product candidates once commercialized, ongoing discussions with potential business partners and expected addressable market size. We cannot assure you that the forward-looking statements in this presentation will prove to be accurate. Some of the factors that could cause actual performance and results to differ materially from those projected or suggested in the forward-looking statements due to various risks and uncertainties, include, but are not limited to, the substantial doubt regarding the our ability to continue as a going concern, our ability to raise additional funding, our ability to enroll patients in future clinical studies, our ability to obtain regulatory approval for our product candidates, unanticipated changes to our nonclinical or clinical study protocols due to regulatory reasons or unanticipated events, which could lead to increased costs to us and could delay our development timeline, our reliance on third parties to conduct clinical studies and to manufacture drug supplies for our product candidates, our ability to adequately protect our intellectual property, and the other important risk factors discussed under the heading “Risk Factors” in our Annual Report on Form 10-K filed with the SEC on March 4, 2025 as well as other risks and uncertainties which may be described in any subsequent quarterly report on Form 10-Q filed by the Company and the other reports the Company files with the Securities Exchange Commission. In light of the significant uncertainties in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified time frame, or at all. The forward- looking statements are made only as of the date of this presentation and the Company undertakes no obligation to update such forward-looking statements to reflect subsequent events or circumstances. Certain market data, study data and industry data used in this presentation were obtained from reports of governmental agencies and industry publications, studies and surveys. Management has not independently verified such data and as such, make no guarantees as to its accuracy, completeness or timeliness.

33 Relaunching Cyclerion: A new, pioneering neuropsychiatric-focused company Strong Company Fundamentals Strategic Pipeline Focus ü Built on opportunity for patient impact and ü Tech-enhanced therapies to optimize patient shareholder value creation benefit ü Driven by lean, nimble team with world-class ü Significant neuropsychiatric market potential neuropsychiatric and biopharma experience with patients requiring new treatment options ü Plan to focus on disciplined advancement ü Novel, improved or first-in-class therapies through de-risked value inflection points ü Validated modes of action with potentially rapid path to clinical Proof-of-Concept (POC) Clear strategic plan to advance programs through de-risked inflection points to drive shareholder value

44 Cyclerion’s neuropsychiatric expertise and agility in action Deep Industry Expertise with Strong Relationships from early research to late commercialization Officers Key Advisors Regina Graul, PhD; CEO Rhonda Chicko; CFO Scholar Rock, Editas, EQRx, Ironwood, Ironwood Cyclerion Laeben Lester, MD Linda Carpenter, Husseini Manji, Anesthesiology leader; MD MD, FRCPC Johns Hopkins Neuropsychiatry Development leader; Board of Directors leader; Brown, Yale, Oxford, J&J, Director of Director NIMH, UK Errol De Souza, Steven Hyman, MD BRaIN*** Govt PhD, Chair Broad Institute of Neurocrine, MIT* and Harvard, Lisa Loram, PhD Lawrence Olanoff, Royalty Pharma Erika De Boever, Provost of Harvard Regulatory Affairs; U of MD, PhD DDS, MPH, PhD NIMH** Director Colorado, Boulder R&D; MUSC, Forest, Clinical Development; Celsion U of Michigan Michael Higgins Peter Hecht, PhD Voyager Tisento, Cyclerion, Therapeutics, Ironwood Michael Madden Andy Parratt Jessica Duda Camp4, Polaris Medical Device New Product BD Advisor; Partners, Ironwood Development; Planning & Red Sky Mountain Medical Commercialization; Regina Graul, PhD Dina Katabi, PhD Karuna, AlgavitaBio EQRx, Ironwood, MIT, Emerald Cyclerion Innovations, MacArthur Fellow *Massachusetts Institute of Technology **National Institute of Mental Health *** Brain Research and Interventional Neurotherapeutics (BRaIN) program at Butler Hospital
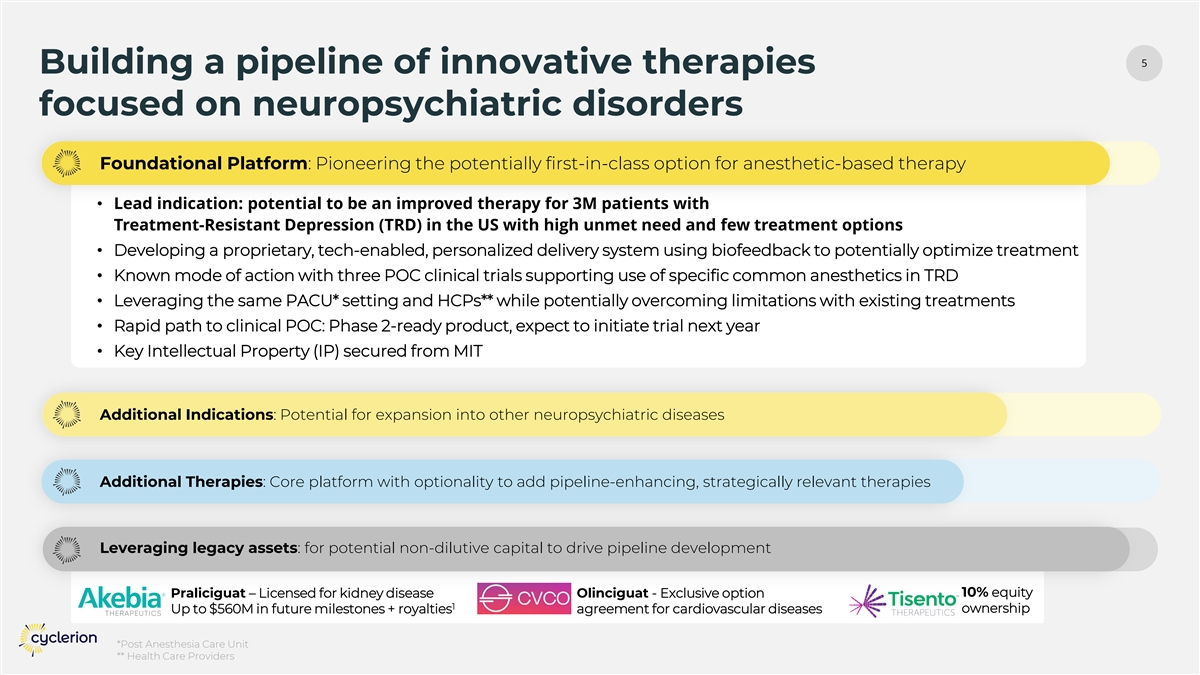
5 Building a pipeline of innovative therapies focused on neuropsychiatric disorders Foundational Platform: Pioneering the potentially first-in-class option for anesthetic-based therapy • Lead indication: potential to be an improved therapy for 3M patients with Treatment-Resistant Depression (TRD) in the US with high unmet need and few treatment options • Developing a proprietary, tech-enabled, personalized delivery system using biofeedback to potentially optimize treatment • Known mode of action with three POC clinical trials supporting use of specific common anesthetics in TRD • Leveraging the same PACU* setting and HCPs** while potentially overcoming limitations with existing treatments • Rapid path to clinical POC: Phase 2-ready product, expect to initiate trial next year • Key Intellectual Property (IP) secured from MIT Additional Indications: Potential for expansion into other neuropsychiatric diseases Additional Therapies: Core platform with optionality to add pipeline-enhancing, strategically relevant therapies Leveraging legacy assets: for potential non-dilutive capital to drive pipeline development Praliciguat – Licensed for kidney disease Olinciguat - Exclusive option 10% equity 1 ownership Up to $560M in future milestones + royalties agreement for cardiovascular diseases *Post Anesthesia Care Unit ** Health Care Providers
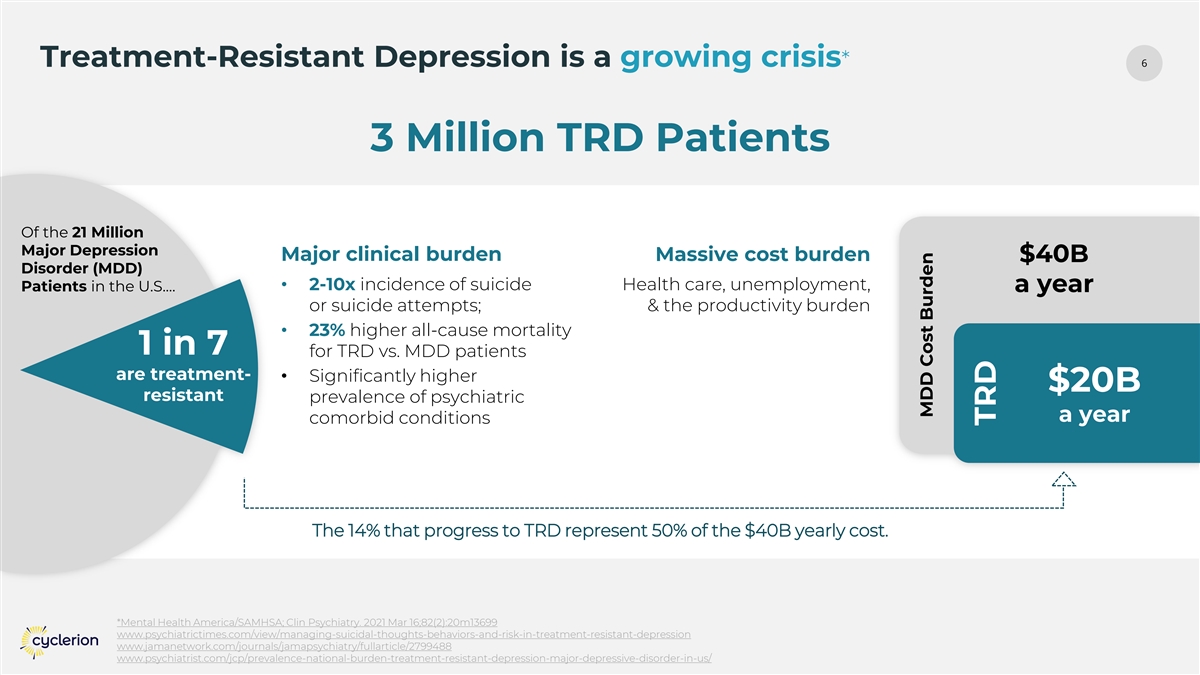
6 Treatment-Resistant Depression is a growing crisis* 3 Million TRD Patients Of the 21 Million Major Depression Major clinical burden Massive cost burden $40B Disorder (MDD) • 2-10x incidence of suicide Health care, unemployment, Patients in the U.S.… a year or suicide attempts; & the productivity burden • 23% higher all-cause mortality 1 in 7 for TRD vs. MDD patients are treatment- • Significantly higher $20B resistant prevalence of psychiatric a year comorbid conditions The 14% that progress to TRD represent 50% of the $40B yearly cost. *Mental Health America/SAMHSA; Clin Psychiatry. 2021 Mar 16;82(2):20m13699 www.psychiatrictimes.com/view/managing-suicidal-thoughts-behaviors-and-risk-in-treatment-resistant-depression www.jamanetwork.com/journals/jamapsychiatry/fullarticle/2799488 www.psychiatrist.com/jcp/prevalence-national-burden-treatment-resistant-depression-major-depressive-disorder-in-us/ MDD Cost Burden TRD

7 The pressing need for better approved treatment options for the 3M patients that progress to TRD Treatments Electroconvulsive Therapy (ECT) ECT + Invasive Therapies Cyclerion Foundational Therapy Candidate Spravato rTMS • Anesthetized patient receiving repeated seizure-inducing treatment ECT at the hospital • Acute and chronic safety concerns, including memory loss**** *see prescribing information for full boxed warning; **Repetitive Transcranial Magnetic Stimulation ***DOI: 10.1002/14651858.CD003493; ****DOI: 10.1192/bjo.2020.17
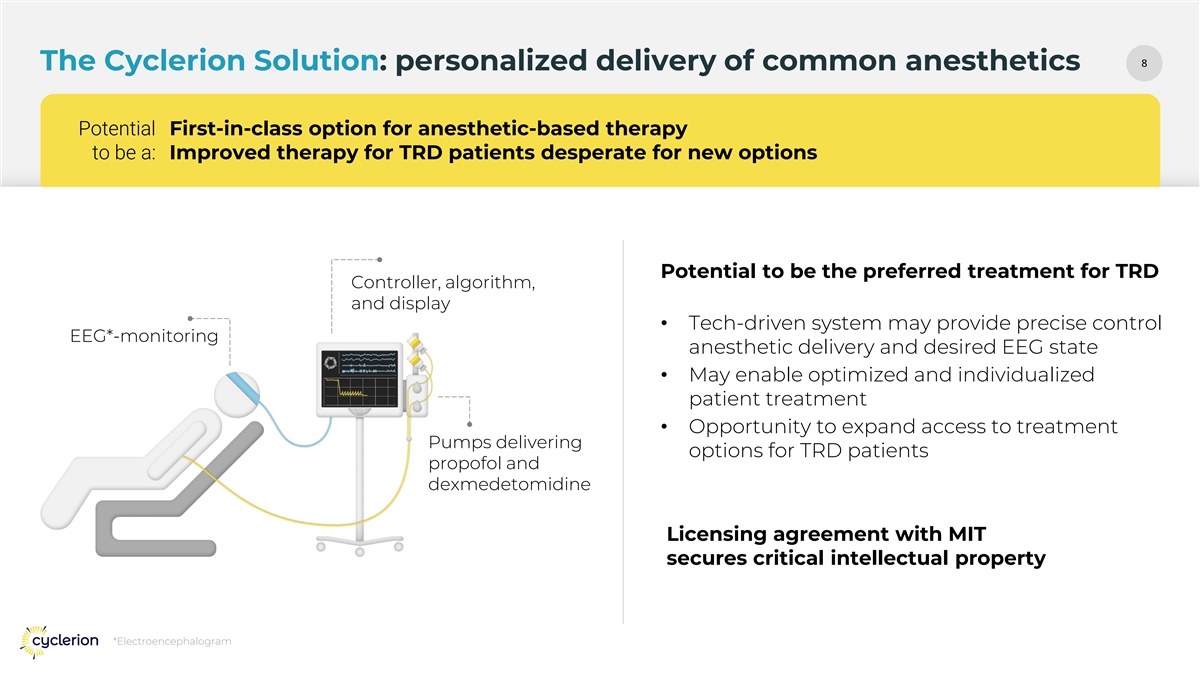
88 The Cyclerion Solution: personalized delivery of common anesthetics Potential First-in-class option for anesthetic-based therapy to be a: Improved therapy for TRD patients desperate for new options Potential to be the preferred treatment for TRD Controller, algorithm, and display • Tech-driven system may provide precise control EEG*-monitoring anesthetic delivery and desired EEG state • May enable optimized and individualized patient treatment • Opportunity to expand access to treatment Pumps delivering options for TRD patients propofol and dexmedetomidine Licensing agreement with MIT secures critical intellectual property *Electroencephalogram
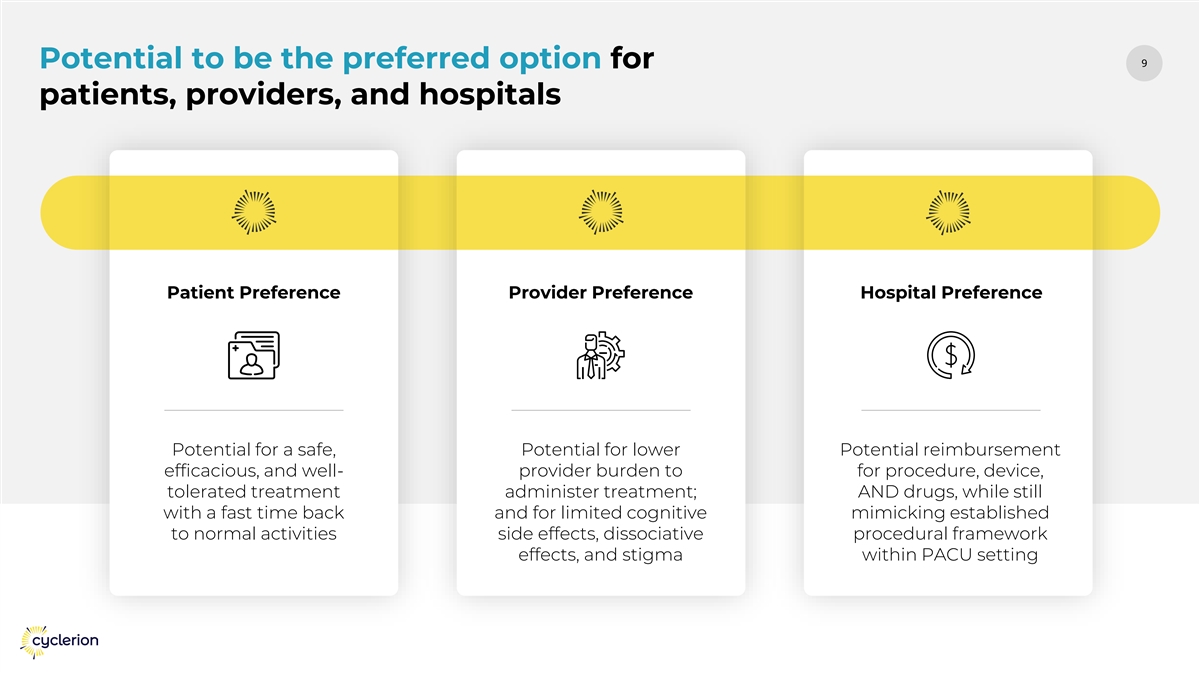
99 Potential to be the preferred option for patients, providers, and hospitals Patient Preference Provider Preference Hospital Preference Potential for a safe, Potential for lower Potential reimbursement efficacious, and well- provider burden to for procedure, device, tolerated treatment administer treatment; AND drugs, while still with a fast time back and for limited cognitive mimicking established to normal activities side effects, dissociative procedural framework effects, and stigma within PACU setting

10 Proven mode of action for use of sedation for TRD Brain regions communicate with each other via oscillatory brain waves: Patient with TRD Cyclerion Therapy Healthy Patients Oscillations Synchronized Specific states of are not oscillations general anesthesia synchronized could facilitate resynchronized communication Communication disrupted between Regions of the brain between key regions key regulating regions of the brain communicating properly of the brain* Improvement in TRD symptoms Select: TRD Symptoms: hopelessness, ruminations, negative thoughts, difficulty regulating emotions, and anxiety *DOI: 10.1093/ijnp/pyy085

11 11 Compelling Clinical Precedent for such use of anesthetics for TRD Three early-phase clinical studies support propofol’s potential as a rapid-acting antidepressant in TRD Anesthesiologist-controlled open loop dosing Propofol showed consistent signals of efficacy with favorable safety profiles was central to all propofol studies: • Rapid onset of antidepressant effect seen • Burst suppression (RCT* + Pilot) or slow-wave sleep (SWS) optimization (SWIPED**) within 1-2 weeks • Propofol titrated using EEG metrics • Durable benefit lasting 3–6 months (e.g., suppression ratio, SWS density) • No major safety signals *Randomized Control Trial **DOI: 10.1136/bmjopen-2024-087516

12 12 Compelling Clinical Data for such use of propofol for TRD Propofol dosing and enhancement of slow wave sleep predict antidepressant response in geriatric patients with treatment-resistant depression [Poster presentation]. Palanca, B. J. A., Ching, S., Farber, N. B., Lin, N., Lucey, B. P., Reynolds, C. F., Lenze, E. J., & SWIPED Study Team. (2025). Gordon Research Conference on Depression, Ventura, CA. Population and Design • N=24: moderate-to-severe TRD patients • N=15 geriatric (≥ 60) TRD patients • N=12: high dose to suppress EEG activity • 2 × 2-hour infusions, 2–6 days apart • N=10: moderate-to-severe TRD patients • N=12: low dose to avoid BS • Manually dosed to avoid burst • 10 propofol infusions over 3 weeks • N=8: crossed over from low dose to high dose suppression ~2 hr infusion • 6 propofol infusions (3x/week for 2 weeks) Results High Dose • 50% (6/12) responders; 42% (5/12) remitters • 67% (10/15) responders • 60% (6/10) responders Low Dose • Dose-dependent percentage change in • 50% (5/10) remitters • 8% (1/12) responders; 8% (1/12) remitters baseline MADRS at 3 weeks: correlates • 4/5 remitters sustained remission ≥3 months; inversely with average propofol dose Crossed Over Low Dose to High Dose 3 of which continued to ~5 months, 2 of administered • 50% (4/8) responders; 42% (3/8) remitters which continued to >6 months Additional Details • Efficacy ~1 week after final infusion; HRDS assessments up to 2 weeks • Efficacy ~2 days after final infusion; HRDS assessments up to 6 months • Responder = ≥30% reduction from baseline MADRS/ • Responder = ≥50% reduction from baseline on MADRS > 6 change in MADRS at any point • Responder = ≥50% reduction from baseline on MADRS • Remitter = Final HDRS ≤10 • Remitter = Final HDRS ≤10
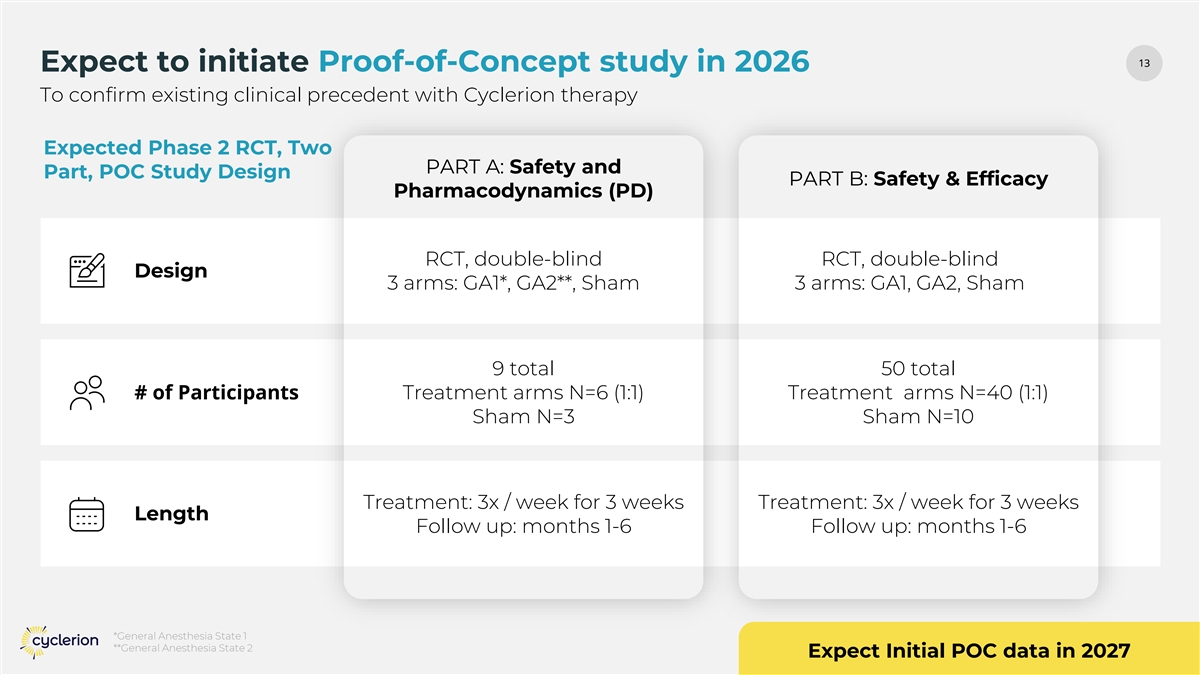
13 13 Expect to initiate Proof-of-Concept study in 2026 To confirm existing clinical precedent with Cyclerion therapy Expected Phase 2 RCT, Two PART A: Safety and Part, POC Study Design PART B: Safety & Efficacy Pharmacodynamics (PD) RCT, double-blind RCT, double-blind Design 3 arms: GA1*, GA2**, Sham 3 arms: GA1, GA2, Sham 9 total 50 total # of Participants Treatment arms N=6 (1:1) Treatment arms N=40 (1:1) Sham N=3 Sham N=10 Treatment: 3x / week for 3 weeks Treatment: 3x / week for 3 weeks Length Follow up: months 1-6 Follow up: months 1-6 *General Anesthesia State 1 **General Anesthesia State 2 Expect Initial POC data in 2027
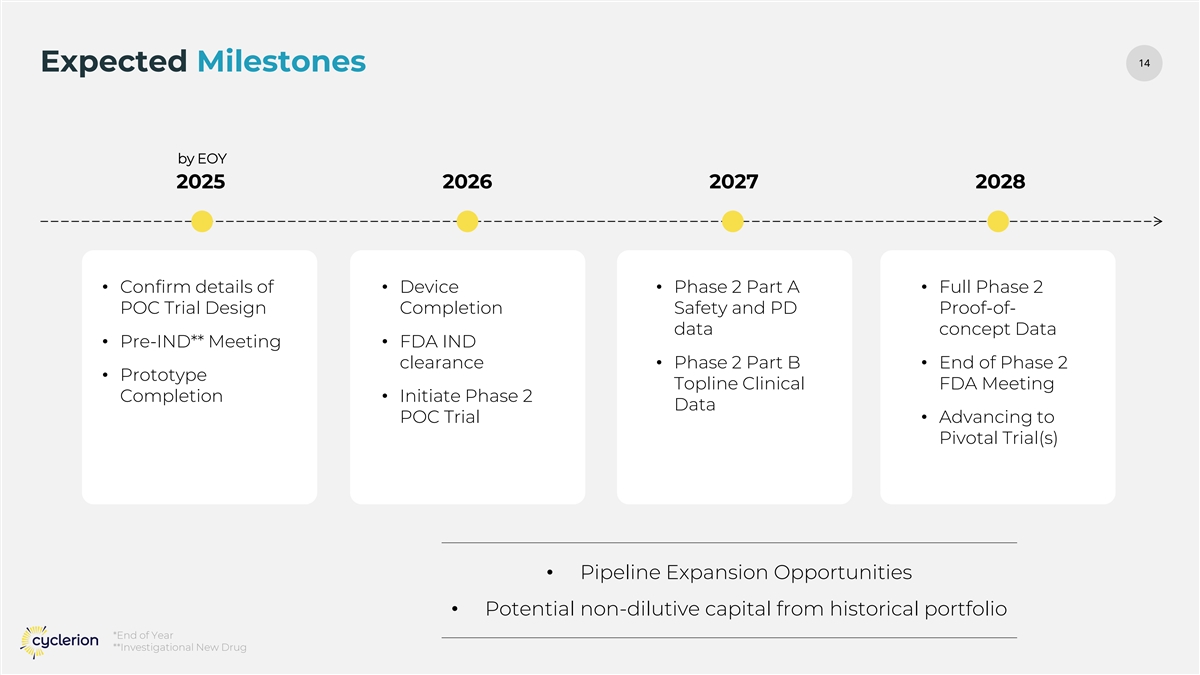
14 14 Expected Milestones by EOY 2025 2026 2027 2028 • Confirm details of • Device • Phase 2 Part A • Full Phase 2 POC Trial Design Completion Safety and PD Proof-of- data concept Data • Pre-IND** Meeting • FDA IND clearance • Phase 2 Part B • End of Phase 2 • Prototype Topline Clinical FDA Meeting Completion • Initiate Phase 2 Data POC Trial • Advancing to Pivotal Trial(s) • Pipeline Expansion Opportunities • Potential non-dilutive capital from historical portfolio *End of Year **Investigational New Drug

Thank You September 2025
